Diffusion
Concentration Gradient
Substances can move across cell membranes. This is how substances are transported into and out of cells. The rate in which substances can move across the cell membrane will depend on the concentration gradient. If there are more particles within a specific volume, those particles are considered ‘concentrated’. A solution will have a low concentration of solute if there is a high water potential. There is a concentration gradient when there is a region of high concentration, and a region of low concentration.
It is considered going down the concentration gradient if the substance is going from high to low concentration.
It is considered going against the concentration gradient if the substance is going from low to high concentration.
Importance of Substance Transportation
It is vital for substances to be transported in and out of a range of organisms. These substances include oxygen, carbon dioxide, water, dissolved food molecules, mineral ions and urea. Cells must remove waste substances and to take in useful substances in order for the cell to function. These substances exchange through the cell membrane through diffusion, osmosis and active transport.
Surface Area Volume Ratio
Multicellular organisms require exchange surfaces and a transport system in order for substances to pass in and out of the cells. The larger the surface area is to the volume, the quicker and more substances can pass through the membranes. Multicellular organisms have specialised exchange surfaces - such as lungs, gills or villi in the intestines so that there is a larger surface area for the transport of substances to take place.
Small and thin structures have a larger surface area compared to the volume.
Large and thick structures have a smaller surface area compared to the volume.
Surface area = Number of sides x (length x length)
Volume = length x length x length
So, as the volume increases, the surface area does not increase at the same rate. If there was a sugar cube that was 5g, it will have a larger SA:V ratio to 5g of powdered sugar.
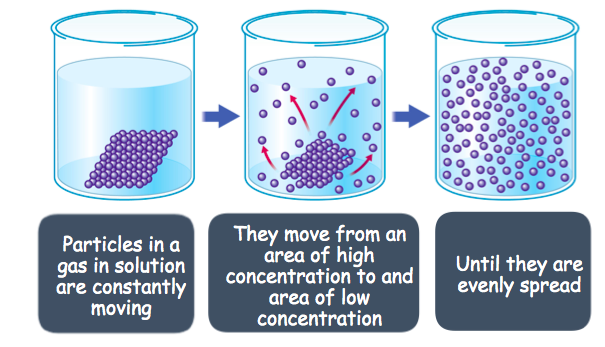
Diffusion
Diffusion is when particles spread from an area of high concentration to an area of low concentration. The molecules would move randomly, and they would eventually become evenly distributed. The particles move down the “concentration gradient” and this process takes no energy (it’s a “passive” process”). Diffusion can be accelerated by increasing the temperature of the particles, which makes them move faster.
Examples of diffusion:
- When someone sprays perfume on themselves, but the smell ends up spreading across the room.
- When placing potassium permanganate crystals into beaker full of water, the entire beaker of water will turn purple
- Products of digestion are normally dissolved in water, which then pass through the small intestine walls
- Oxygen moving down the concentration gradient as it moves from the air in the alveoli into the blood
- Carbon dioxide moving down the concentration gradient from the blood to the air in the alveoli
Rate of Diffusion
The following factors have an affect on the rate of diffusion:
- Surface Area
The larger the surface area to volume ratio, the quicker the rate of diffusion takes place. The substance simply has more area to diffuse across, despite being the same volume.
- Concentration Gradient
The higher the concentration differences in the gradient, the higher the diffusion rate. If there is an equal concentration on both sides of the membrane, the rate of diffusion will be extremely slow. E.g. if the alveoli has a much higher concentration of oxygen than in the blood, then the oxygen will diffuse into the blood much quicker.
- Diffusion Distance
The shorter the distance between the material in which the substance is diffusing through, the faster the diffusion rate. It simply has a shorter journey to travel.
- Temperature
By increasing the temperature, the rate of diffusion increases as there is more energy in each particle. These particles will now bounce against each other more frequently.
Diffusion in Multicellular Organisms
In multicellular organisms, surfaces and organ systems are specialised for exchanging materials. This is to allow sufficient molecules to be transported into and out of cells for the organism’s needs. Small intestines, lungs (in mammals), gills in fish, roots and leaves in plants are all adapted for exchanging materials, as its exchange surface is increased by:
- Having a large surface area
- A membrane that is thin, to provide a short diffusion path
- (in animals) Having an ef cient blood supply
- (in animals, for gaseous exchange) Being ventilated
- Name 4 factors that can affect the rate of diffusion.
- Your answer should include: Temperature / Diffusion / Distance / Concentration / Gradient / Surface / Area
- When there is an increase in temperature, why is there an increase in the rate of diffusion?
- Your answer should include: Particles / Energy / Bounce
- Is diffusion an active or passive process?
- Passive