Properties of Radiation
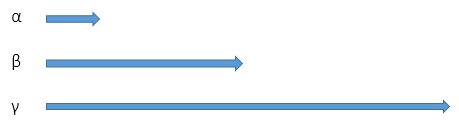
Range
Alpha can travel a few centimetres in air.
Beta can travel a few metres.
Gamma rays have an almost infinite range.
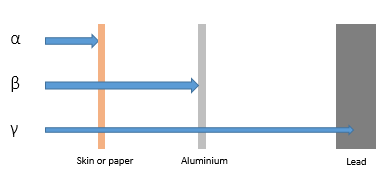
Penetration
Alpha can be absorbed by paper or skin.
Beta can be absorbed by thin aluminium.
Gamma rays can be absorbed by thick lead.
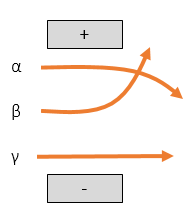
Magnetic and Electric Fields
Gamma rays will pass through a field unaffected as they have no charge and no mass.
Beta will be deflected towards the positive area of the field. It has a low mass so will be strongly deflected.
Alpha will be deflected towards the negative area of the field. It has a high mass so will be weakly deflected.
###
Danger
Which type of radiation is the most dangerous depends on the situation. Alpha is the most ionising but is least likely to reach you due to it’s short range and low penetration. Gamma is the least ionising but is the most likely to reach you due to it’s long range and high penetration.
You can be exposed to radiation in two ways:
- Irradiation: Exposure to a radioactive source outside of your body
- Contamination: Exposure to a radioactive source inside your body
Half Life
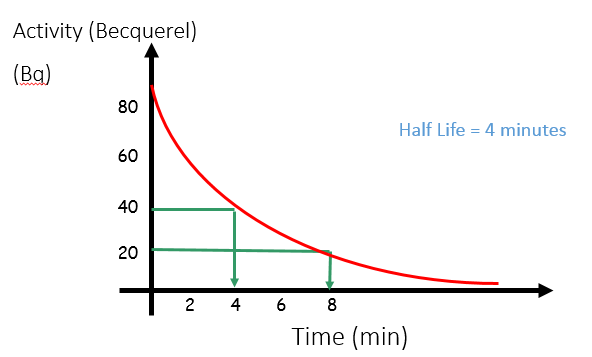
The half-life is the time it takes the (radio)activity of a substance to decrease by half.
Half-life can be calculated from a graph of the activity level of a radioactive substance over time. At any point on the graph (but it’s usually easiest to do it from the start), work out the time taken for the activity to drop by half – this is the half-life.
If the half-life is 4 minutes, then every 4 minutes, the activity will decrease by half.
Half-life can also be calculated numerically.
For example:
A sample of Bismuth-214 has an activity of 64Bq. It has a half-life of 20 minutes.
- After 20mins (1 half-life), the activity will be 32Bq (half the original activity
- After 40mins (2 half-lives), the activity will be 16Bq (half again)
- After 60mins (3 half-lives) the activity will be 8Bq
And so on.
Ionisation
Ionising radiation has enough energy to move electrons from atoms. This is harmful to cells in your body (cancer). Ionising radiation can also be used to kill cancerous cells.
Alpha is the most ionising.
Gamma is the least ionising.
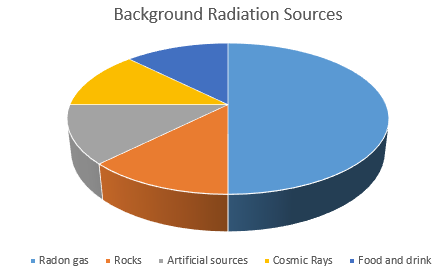
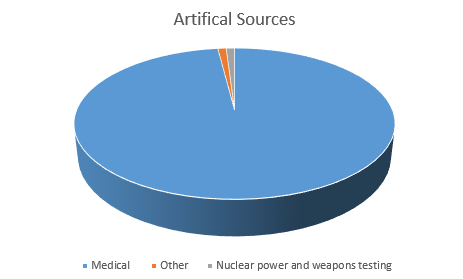
Background Radiation
Some elements emit ionising radiation all the time. These radioactive elements are naturally found in the environment. Background radiation includes natural and man-made radioactive sources.
Exposure to radiation can happen in two ways:
- Irradiation - Exposure to radiation from a source outside your body
- Contamination - Exposure to radiation from a source inside your body
Carbon Dating
Living things contain some radioactive carbon-14. When the organism dies, it stops taking in carbon-14. The carbon-14 decays to carbon-12. The half-life of carbon-14 is 5600 years. The level of radiation emitted from the carbon 14 can be used to work out how long ago the organism died.
Worked Example
A living bone emits 170 counts per minute. A bone from an archeological site emits 50cpm. The background radiation in the area is 10 cpm. How old is the bone? (Half-life of Carbon = 5600years)
- Fresh bone – background radiation = 170 – 10 = 160cpm
- Ancient bone – background radiation = 50 – 10 = 40cpm
- Fresh = 160cpm. 1 half-life later = 80cpm. After 2 half-lives = 40cpm
- The ancient bone is 2 half-lives old.
- Half-life = 5600 years
Ancient bone = 120,000 years old
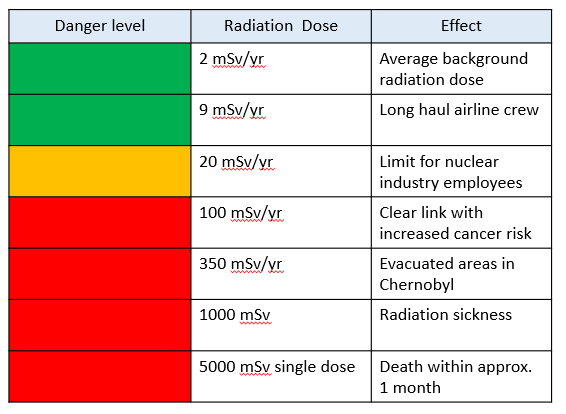
Safety Precautions
The risk to general public from radioactive materials is low, but people working with radioactive substances need to take precautions.
They can avoid contamination by:
- Careful storage of radioactive materials
- Wearing gloves/protective clothing
They can reduce irradiation by:
- Working behind lead shields/barriers
- Reducing time in at risk areas
Monitor exposure using a film badge
Using Radiation
When using radioactive materials, the properties of the source need to be matched to the specific need of that task considering:
- Range
- Penetration
- Ionisation
- Half-life
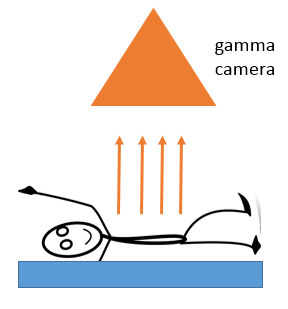
Medical Diagnosis - Gamma Camera
To use a scan from a gamma camera, the patient swallows a gamma tracer or has it injected into the bloodstream. The camera detects the gamma radiation emitted from the body, producing an image of the body parts containing the tracer.
A gamma source is used as it is the least ionising and will pass through the body easily to be detected by the camera. It is important to choose a half-life which is long enough to allow the scan to be carried out but not so long that the patient remains contaminated for a long time.
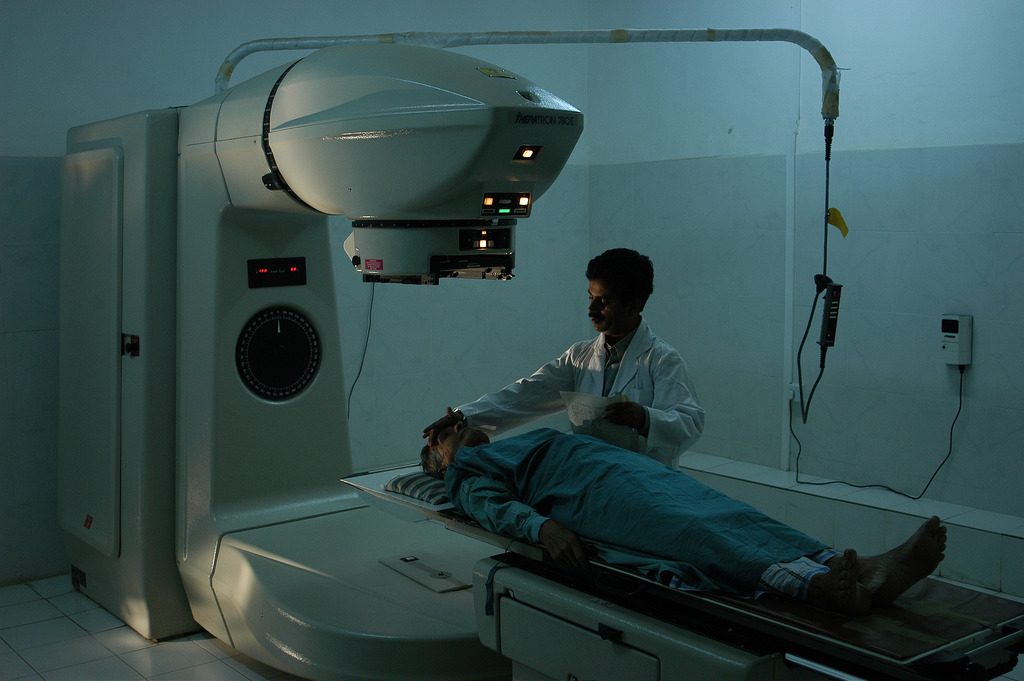
Medical Treatment – Radiotherapy
As ionising radiation is dangerous to cells, it can be used to kill cancerous cells. The radiation is targeted at the tumour, but some damage to healthy cells is inevitable. Gamma or beta sources are usually used as alpha cannot penetrate the skin.
Sterilisation
Radiation can be used to kill bacteria and therefore sterilise surgical equipment. The items can be sealed in air-tight packaging before being irradiated, which means there is no chance of re-contamination before they are used.
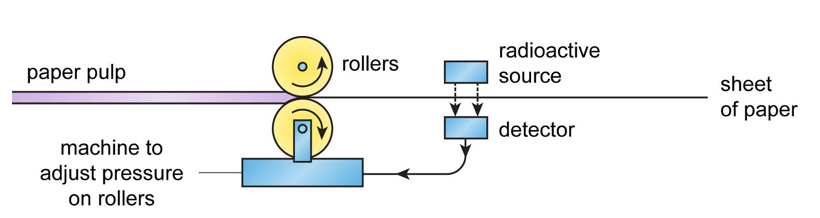
Measuring Thickness
Radioactive sources can be used to measure the thickness of materials that are being rolled out, such as paper or sheet metal. The source is positioned on one side of the material with a detector on the other side. A change in thickness will affect the amount of radiation passing through the material and being received by the detector. This can be used to change the pressure of the rollers to increase/decrease the thickness as necessary.
Beta is used for paper (as alpha can not pass through paper and gamma would pass through too easily). Gamma is used for metal as alpha and beta can not pass through.
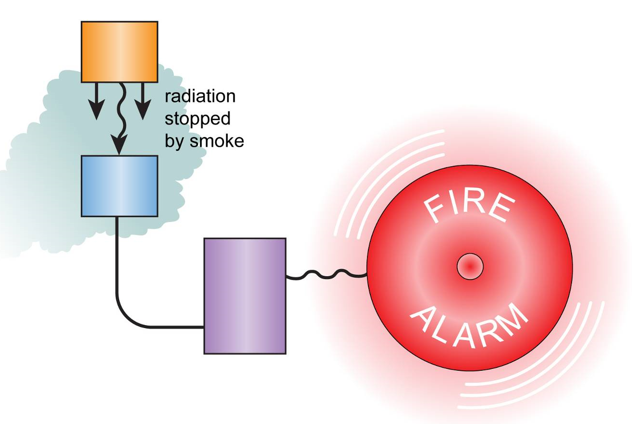
Smoke Detectors
Smoke detectors use an alpha source which ionises the air in a small gap in a circuit, allowing current to flow.
If smoke is present, it absorbs the alpha radiation meaning current can now no longer flow across the gap, this triggers the alarm.
An alpha source is used because it can not penetrate through smoke. Beta and Gamm would not be suitable as they can pass through smoke.
- Which type of radiation is the most ionising?
- alpha
- Which type of radiation has no electrical charge?
- gamma
- Which type of radiation has the largest range?
- gamma
- The activity of a radioactive sample is 1440Bq. 5 hours later it is 45Bq. What is the half life of this sample?
- Your answer should include: 1 hour / 1
- The half life of Strontium-90 is 29 years. If you start with 1000 nuclei of Strontium-90, how many would you expect to be left after 87 years?
- 125