Enzymes and Digestion
Enzymes
Millions of chemical reactions are happening in our body at all times. A chemical reaction can normally be made faster by increasing the temperature. However, this also would increase the speed of other reactions (which we might not want to happen). So… our body uses biological catalysts instead!
- Enzymes are biological catalysts!
- A catalyst is something that speeds up a reaction and is the same after the reaction as they were at the start of the reaction.
- Enzymes (biological catalysts) reduce the need for high temperatures in the body.
- Enzymes are large proteins folded in a unique way. (Proteins are chains of amino acids!)
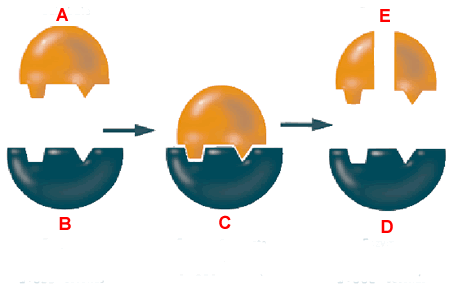
Active site:
There are lots of different types of enzymes. They all have a uniquely shaped active site. The active site of a protein is the site in which all the magic happens!
The active site of an enzyme is a unique shape and can only bind with substrates that have a compatible shape.
If the substrate doesn’t fit in the active site, then the enzyme can’t catalyse the reaction.
This shows the ‘lock and key’ model of an enzyme. It shows that the substrate (A) needs to fit in the active site of the enzyme (B) in order to be broken up (E).
Denaturing Enzymes
If enzymes are not in the right conditions (usually temperature and pH) then the active site can deform its shape! If the active site is deformed then it won’t fit with any substrate and won’t be able to catalyse any reaction (and will be useless).
Once an enzyme has become denatured, it cannot return back to normal.
Enzymatic Reactions
Optimum pH and temperature
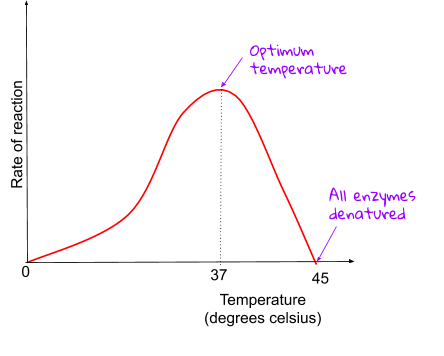
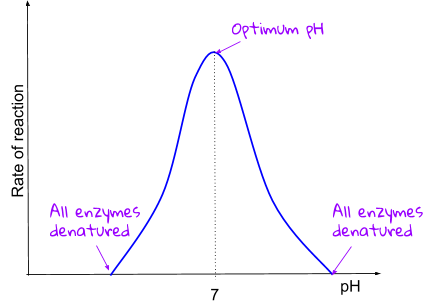
Enzymes work the best when they’re at their optimum pH and temperature. Optium just means best, so enzymes work best at certain pH’s and temperatures. However, not all types of enzymes like the same pH or the same temperature!
Increasing the temperature increases the rate of reaction, but if the temperature gets too high, enzymes start to denature (change shape) as the bonds holding the enzyme together start to break.. The more that are denatured, the slower the rate of reaction. Enzymes in the body usually have an optimum temperature of 37℃, but this isn’t always the case!
The wrong pH can also break some of the bonds holding the enzyme together, changing she shape of the active zone and hence denaturing them. The optimum pH for enzymes is different (this is why we have stomach acid). But some enzymes have an optimum pH around 7.
Investigating the effect of pH and temperature on enzyme action
You can easily investigate the effects of pH and/or temperature on enzymes. There will be more on this in the FoodTests section.
Enzymes and Digestion
The digestive system is the system of organs responsible for breaking down and digesting food.
There are two ways in which food is broken down in the body:
- Mechanical digestion - which includes grinding down food with teeth or churning up food with stomach muscles.
- Chemical digestion - which includes breaking down food through enzyme action (enzymes breaking them up into smaller pieces).
There are three main types of enzymes that work in your digestive system:
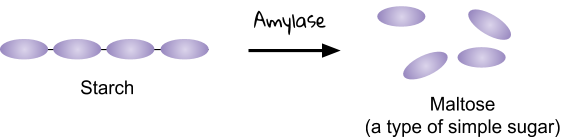
Carbohydrase - breaks down carbohydrates into simple sugars. For example, amylase (a type of carbohydrase) breaks starch into maltose sugar.
__Protease __- breaks down protein into amino acids.
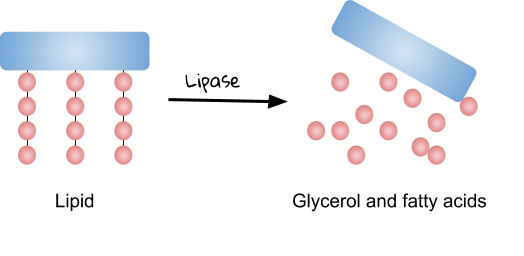
__Lipase __- breaks down lipids (fats) into fatty acids and glycerol.
This table is AMAZING and you definitely should memorise it! It will get you out of a lot of tricky question about the digestive system.
You can read this table like a sentence. For example. Let’s look at Amylase.
The amylase enzyme helps to break down starch into sugars. This enzyme is made in the salivary glands, pancreas and small intestine, and works in the mouth and small intestine.
| Enzyme/Enzymes | Amylase | Proteases | Lipases |
| helps to break down... | Starch | Proteins | Lipids |
| … into … | Sugars | Amino acids | Glycerol and fatty acids |
| This enzyme is made in the … | Salivary glands, pancreas and small intestine | Stomach, pancreas and small intestine | Pancreas and small intestine |
| This enzyme works in the... | Mouth and small intestine. | Stomach and small intestine | Small intestine |
Optimum p H within the Digestive System
Different enzymes in the digestive system work at different pH’s. For example, protease in the stomach has a very low optimum pH. This is why our stomach is filled with acid, do decrease the pH for the enzyme!
The enzymes in the small intestine like slightly higher pH’s, so something has to be done to increase the pH of the food that is all covered in stomach acid! Here is where bile comes in (bile is that horrible tasting stuff that comes out after you have nothing else to sick up!)
Bile:
- Produced in the liver
- Stored in the gallbladder
- Works in the small intestine
- Bile neutralises the stomach acid that the food picked up on its way through the stomach
- Bile also emulsifies fats for faster digestion. Emulsification just means breaking up large globules into smaller globules of fat. Think of when oil is in water, the oil all sticks together in one globule!
Food Tests
You will first need to prepare your food sample before testing for different food types.
__The preparation for food samples is the same at first, so this is what you do… __
- Break down the piece of food you want to test, using a pestle and mortar.
- Put this ground up food into a beaker and add some distilled water.
- Stir the sample with a glass rod. This is to dissolve some of the food.
- Filter the solution using filter paper and a funnel into a test tube. (don’t do this step for lipids, just transfer it into the test tube as it is).
This will leave you with a liquid solution containing some of the food!
To test for each food type, you just need to follow the table below: