Transport in Cells
Diffusion
What is diffusion?
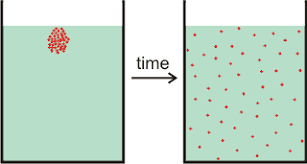
Diffusion is the spreading out of particles, from an area of__ high concentration__ (where there are lots of them) to an area of low concentration (where there are few of them).
Diffusion happens in liquids and gases, since the particles are free to move around! The particles move around randomly and after a little while, the particles are all spread out!
Diffusion in cells
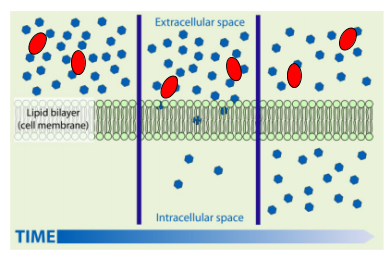
Cell membranes control what goes in and out of the cell. This can happen by __diffusion __or active transport (we’ll talk about this later).
Small particles can fit through the cell membrane but large particles can’t get through! Small particles get through via diffusion. Lets use oxygen particles as an example. If there are lots of oxygen particles on the outside of the cell (like the animation above), the particles will move randomly around (even through the cell membrane). There will be a net movement* of oxygen particles into the cell until there is an equal amount of particles in and out of the cell.
Note: *Net movement means the general movement of particles. So even though the particles are randomly moving into and out of the cell, there is a net movement of particles into the cell.
Rate of diffusion
The speed at which diffusion happens changes depending on a few things.
Temperature: Diffusion rate increases with temperature increase. The hotter the liquid (or gas) the faster the particles move! If the particles are moving faster, an equilibrium is reached faster!
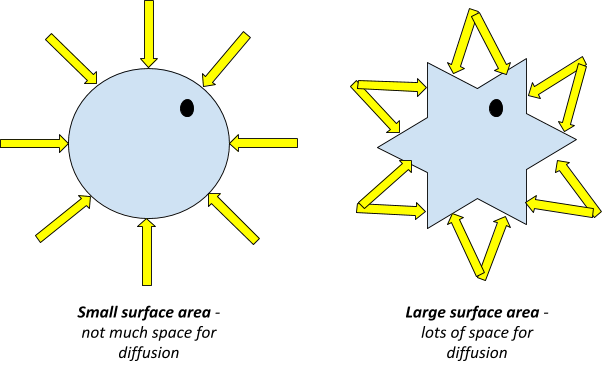
Surface area: The larger the surface area of the cell, the faster the rate of diffusion. Since there are more areas at which particles can travel into and out of the cell.
Concentration gradient: The higher the concentration gradient, the faster the rate of diffusion.
Osmosis
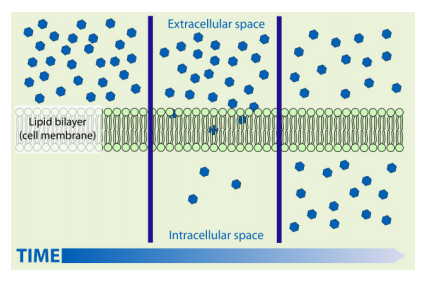
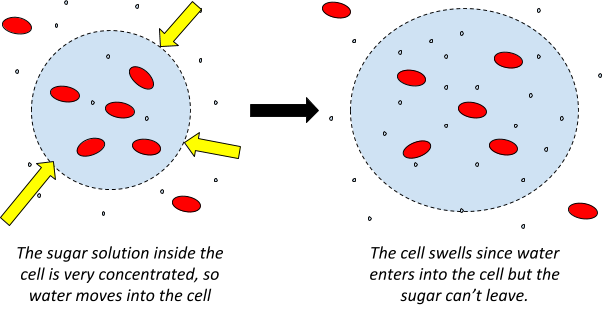
Osmosis is the movement of water particles across a semipermeable membrane, from an area of high concentration to an area of low concentration. It’s just like diffusion but for water!
A semipermeable membrane is just a membrane with tiny holes in it, so that small molecules (like water) can pass through but large particles (like sugar) can’t!
The water molecules are actually moving randomly both ways through the membrane during osmosis. Because there are more water molecules on one side of the membrane, there is a net movement of water into the area of low concentration. You can think of it like the water trying to even up the water concentration on either side of the membrane.
Movement of water into and out of the cell
When a cell doesn’t have enough water but has a lot of sugar, the liquid inside the cell is highly concentrated (where as the liquid outside the cell will have a low concentration). The cell will take up more water (from outside) in order to make the concentration the same inside and outside of the cell.
The opposite happens if there is too much water inside of the cell.
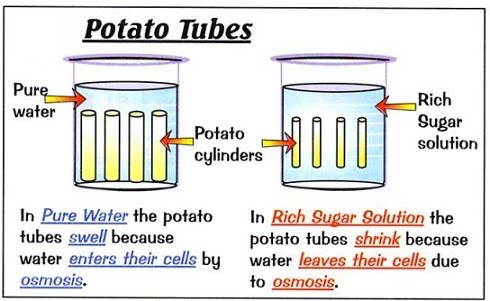
Investigating Osmosis in Potatoes
Method
- Cut the potatoes up into 3 equal size cylinders and measure the mass of each one.
- Make up 2 beakers of sugar solution, both of different concentrations. Make one beaker of pure water.
- Place a potato cylinder into each beaker and leave them for 24 hours.
- Take the potatoes out, dry them and measure the mass of each one again.
Analysing results
If the cylinders have lost water through osmosis then the mass will have decreased. If the cylinders have gained water through osmosis, the mass will have increased.
You should see that the potato that was in the strongest sugar solution has lost the most mass (since it’s lost the most water).
Active Transport
Active transport is the movement of particles from an area of low concentration to an area of high concentration using energy.
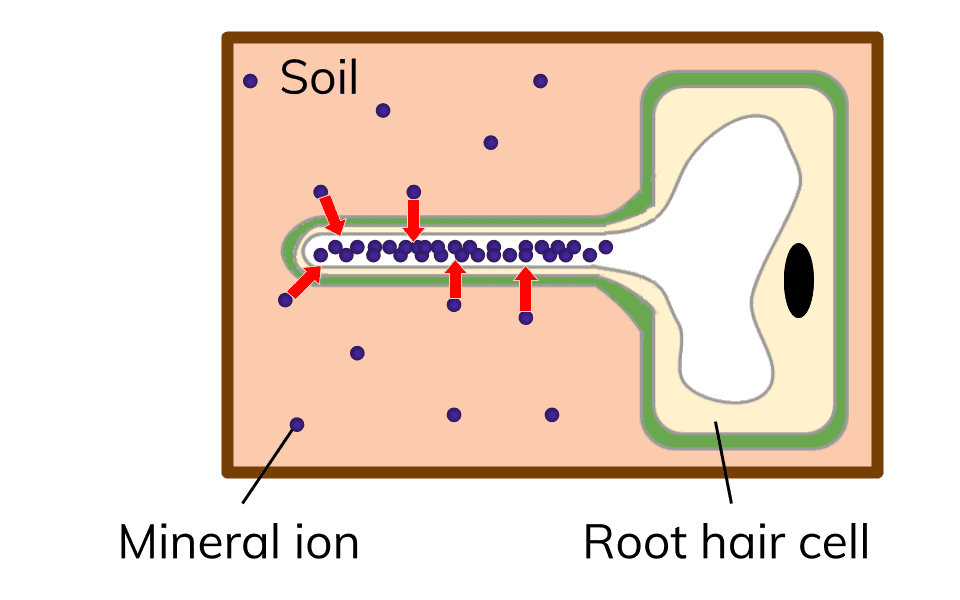
Active transport in plants
Plants need to take up minerals from the soil. However, the mineral ion concentration is usually higher inside the cell then outside the cell. The minerals can’t get into the cell via diffusion, since they would be travelling against the concentration gradient. But they can travel via active transport. Energy is needed to make this happen.
Summary of Transport Methods
| Method | Description |
| Diffusion |
- Passive transport (doesn’t require energy)
- Particles travel from an area of high concentration to an area of low concentration (with the concentration gradient)
- __Small particles travel through the membrane __
| Osmosis |
- Passive transport (doesn’t require energy)
- __Water particles travel from an area of high concentration to an area of low concentration __
| Active Transport |
- __Active transport (requires energy) __
- Particles can travel from an area of low concentration to an area of high concentration (against the concentration gradient)
- __Large particles (and small particles) can travel through the membrane __