Bonding
Metals and Non-Metals
All chemical elements can be divided into either metals or non-metals based on their physical and chemical properties. These properties come from the differences in their atomic structures, especially the arrangement of the electrons.
Physical Properties
| Metals | Non-Metals |
| Lustrous ( shiny) | Dull |
| Good conductors of electricity | Poor conductors of electricity |
| Good conductors of heat | Poor conductors of heat (insulators) |
| High melting point (except mercury) | Variable melting points |
| Solid at room temperature (except mercury) | Can be solid, liquid or gas at room temperature |
| Malleable | Brittle |
| Ductile (can be pulled into a wire) | Non-ductile |
| Opaque in thin sections | Transparent in thin sections |
| Sonorous (make a bell-like noise when hit) | Non-sonorous |
Chemical Properties
| Metals | Non-Metals |
| 1 - 3 electrons in the outer orbit of atoms | 4 - 8 electrons in the outer orbit of atoms |
| Lose electrons | Gain electrons or share electrons |
| Corrode easily (general rule with several exceptions including gold) | |
| Form base oxides (alkaline) | Form acidic oxides |
| Good reducing agents | Good oxidizing agents |
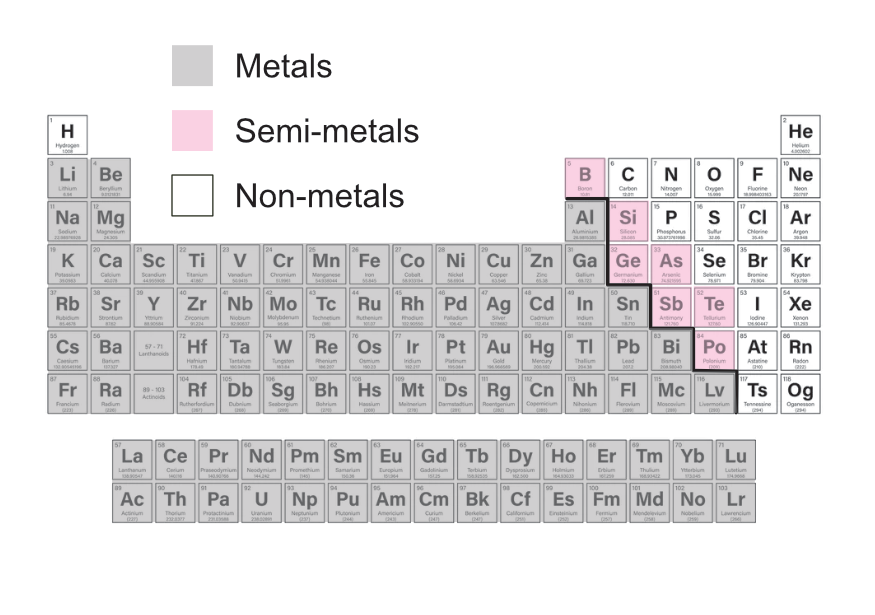
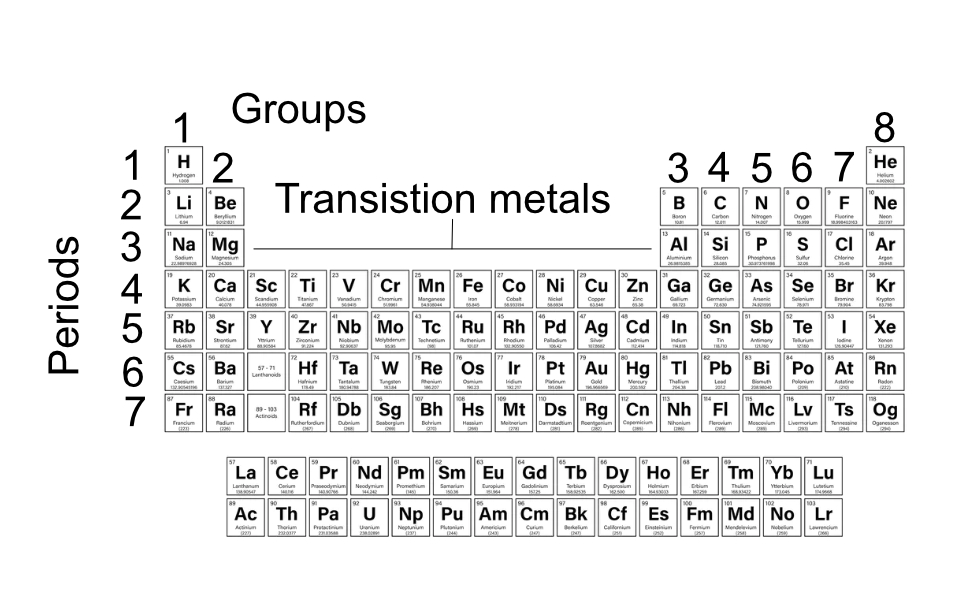
The periodic table arranges elements according to their atomic structure, as such the metals and non-metals are found in different parts of the table as illustrated below.
Some elements have properties at are a mixture of metal and non-metal properties, these are shown as the semi-metals in the table.
Chemical Bonds
Elements without a full outer electron orbit will react with other elements to try and end up with a full outer orbit. There are three ways an atom can do this.
- Remove the electrons in the current outer orbit. (metals)
- Add electrons from another atom to fill the current outer orbit (non-metals)
- Share electrons between atoms to complete the outer orbit (non-metals)
Swapping or sharing electrons between atoms creates the two main types of chemical bonds.
- Ionic Bonds: swapping electrons between metal and non-metals elements.
- Covalent Bonds: sharing electrons between non-metal elements.
Ionic Bonds
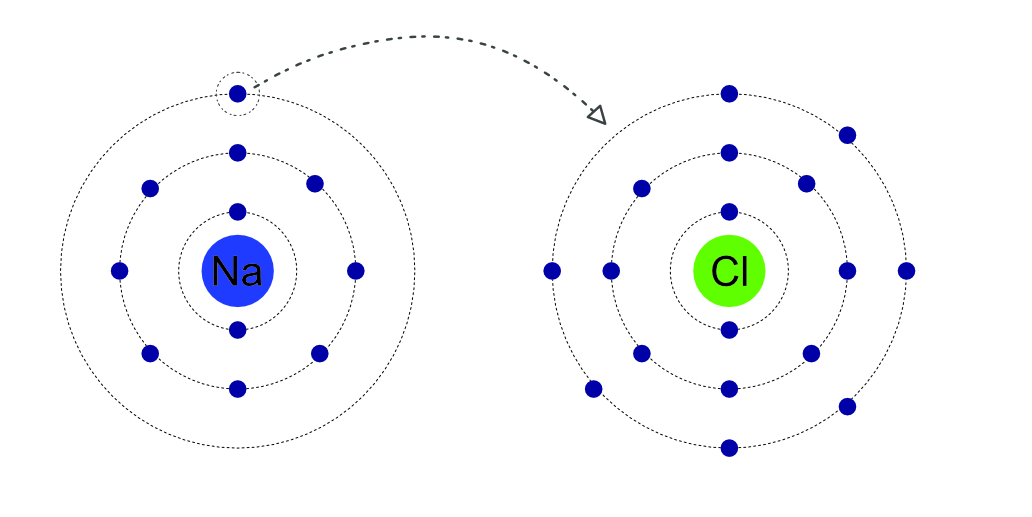
These bonds are formed between a metal and a non-metal elements. The spare electrons in the outer orbit of the metal atom are transferred to the outer orbit of the non-metal. This leaves each atom with a full outer orbit, but as they have gained or lost electrons they are now both electrically charged ions. The metal becomes positive and the non-metal negative. This creates an electrical attraction between them. It is this attraction that forms the bond, there is__no__ physical connection between the atoms only an electrical attraction force.
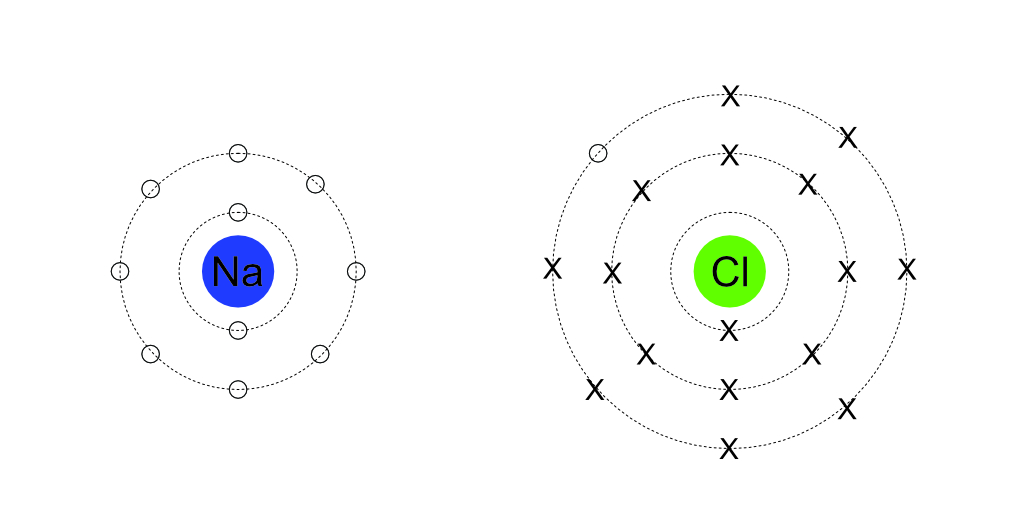
The single electron in the outer orbit of the sodium atom is transferred to the outer orbit of the chlorine atom. This fills the outer orbit of the chlorine. The loss of the electron makes the sodium atom become a positive sodium ion, the additional electron in the orbit of chlorine makes it becomes a negative chlorine ion. The positive and negative attract to form sodium chloride.
Simple Covalent Molecules
The transfer of electrons between metals and non-metal requires energy, to move more than 3 electrons in this fashion is too energetic. Atoms can fill their outer orbits by sharing electrons, effectively merging their outer orbits so that the electrons orbit more than one atom at a time. This arrangement creates a covalent bond and is formed between non-metal elements only.
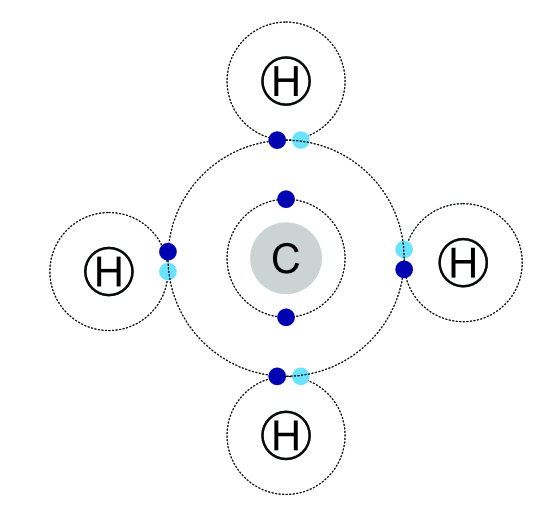
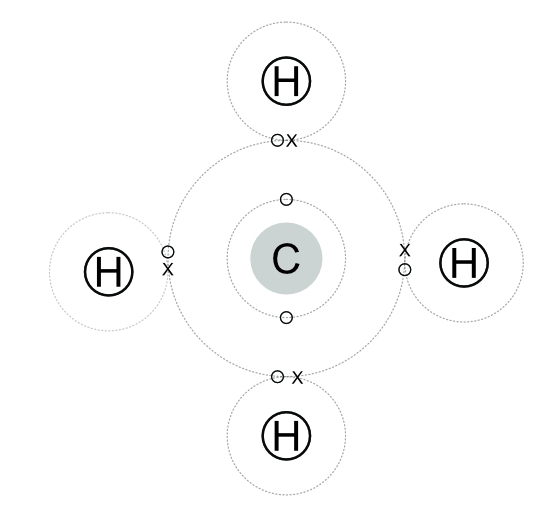
In this methane molecule the single electron in the orbit of each of the four hydrogen atoms orbits around the hydrogen and the central carbon atom. The electrons in the carbon orbit both the carbon and the hydrogens. This combination gives the carbon an outer orbit of eight electrons and each of the hydrogens an outer orbit of two. This gives the effect of each atom having a full outer orbit.
Giant Covalent Structures
Covalent bonds can only form between non-metal elements, they can form very large complex molecules such as diamonds, graphite and organic molecules such as sugars and DNA.
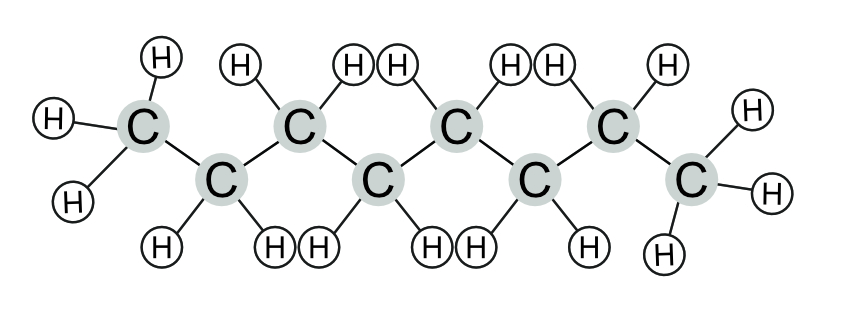
Diamonds are made of pure carbon arranged in a 3-D triangular style shape. Each carbon is held in place by covalent bonds with its neighbouring carbon atoms. Octane is a hydrocarbon made from a chain of carbon atoms, surrounded by hydrogen atoms. All the bonds in this structure are covalent.
Polymers
A polymer is a long chain of repeating units, called monomers. These long complex molecules are held together by a series of covalent bonds. These include organic chemicals such as proteins and non-organic compounds such as polyethene and many other plastics. These structures normally form strong bonds that are very stable.
Metal Bonds
Unlike the covalent bonds between non-metals the bond that holds a metal structure together are less strong. The electrons in a metals outer orbit are only held in place by a weak force from the nucleus. In a metal material, the electrons move from one metal atom to another. They are shared between the surrounding atoms. This creates the effect of the nuclei sitting in a sea of free moving electrons. This is why metals can conduct electricity.
Metals nuclei sit in a “sea” of free moving electrons forming weak metallic bonds.
Periodic Table
The periodic table, originally constructed by Dmitri Mendeleev arranges all the known elements according to their atomic structures. The position of an element can provide information on the number of electrons, the number of orbits and most importantly the number of electrons in the outer orbit of the atom. This is important because this affects whether an element is a metal or a non-metal and how it reacts with other elements.
Groups 1 to 8 shows the number of electrons in the outer orbit of the atoms and the periods indicates the number of orbits of electrons.
Examples: Lithium is in group 1 and period 2, so it has 2 orbits and the outer orbit has 1 electron. Chlorine is in group 7 and period 3. It, therefore, has 3 orbits and the outer orbit contains 7 electrons.
The modern periodic table is based in the work of Dmitri Mendeleev who published the first periodic table in 1869. He organised the elements according to their atomic mass and realised that many of their chemical properties were related to their relative atomic mass. He also recognised that the pattern in his table was incomplete and that there must be other elements that had not yet been discovered. Over time these have been added and the structure of the table adapted.
Dot and Cross Diagrams
Dot and cross diagrams are used to represent simple bonds between atoms. The electrons in one atom are drawn as an “x” and the other electrons of the other atom as a dot. This helps to illustrate which electrons have moved or are being shared between atoms.
In this methane molecule the electrons of the carbon shown as an “O” and those of the hydrogen as an “X”.
In this ionic example, one of the “O” representing the electrons of the sodium atom is in the outer orbit of the chlorine. The sodium ion has one less electron and one less orbit than a sodium atom.
Like all models these are useful to help illustrate the process involved in forming bonds, however, they have some limitations. The models do not give a full picture of the three-dimensional nature of molecular structures, nor can they show the strength of the bonds and the forces between atoms with a molecule.