Preparation of Salts
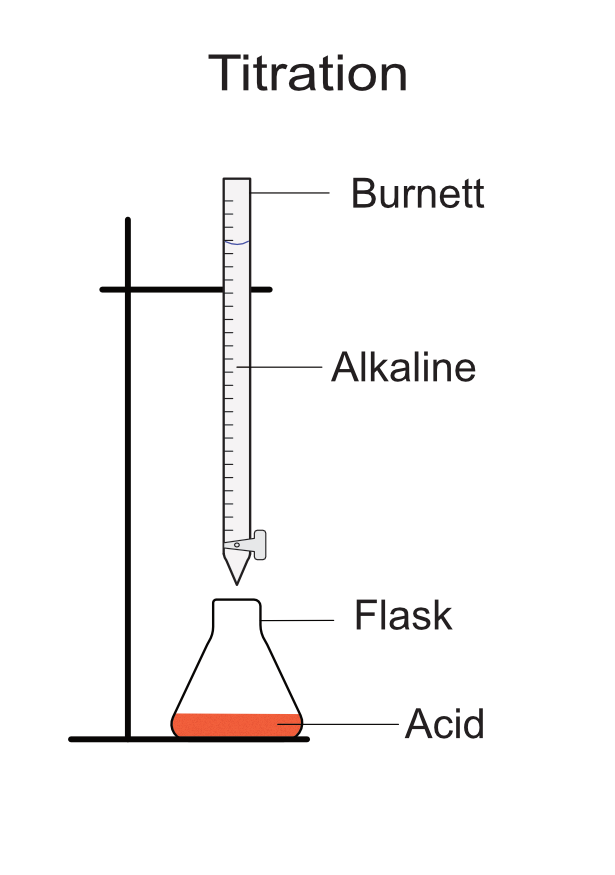
Titration
Salts are made from the neutralization of an acid with either a metal, an alkaline or a carbonate.
Titration is a method for accurately measuring the volume of acid and an alkaline required to produce a neutral solution. This gives the quantities of chemicals required to completely neutralize the acid and the alkaline and provide the maximum amount of a salt.
Safety:
1M Sodium hydroxide: Corrosive: handle with care - wear goggles at all times.
1M Hydrochloric acid: Corrosive: handle with care - wear goggles at all times.
Reaction: Sodium hydroxide + Hydrochloric acid → Sodium chloride + Water
NaOH(l)+ HCl(l) → NaCl(aq) + H2O(l)
Equipment:
- 100 ml 1M HCl
- 100 ml of 1M NaOH
- Measuring cylinder 100ml or graduated pipette and bulb
- Flask
- 50 ml burette
- Funnel
- 50 ml beaker
- Clamp stand
- Universal indicator
- Dropper pipette
- A sheet of white paper
Method
- Place 25 ml of 1M HCl into the flask using the graduated pipette or measuring cylinder.
- Place the burette into the clamp stand and ensure the tap is CLOSED.
- Fill the burette with 1M NaOH using a funnel. - Add a small amount at first with an empty beaker below the tap to ensure it is not leaking.
- Add two or three drops of universal indicator to the acid in the flask and mix gently.
- Place the flask below the burette.
- Open the tap slowly to add a few drops of NaOH to the HCl. Stir the mixture. Continue this slowly until the indicator turns green. A sheet of white paper can be placed below the flask to see any colour change if needed.
- Record the volume of NaOH used to neutralize the HCl.
A fresh sample of 25 ml 1M HCl can now be combined with the correct volume of NaOH without using the indicator to obtain a neutralized solution of NaCl(aq) or sodium chloride solution.
Set up:
Evaporation
In order to obtain a dry sample of the salt from the titration, the NaCl must be separated from the water. There are two methods to do this.
- The salt solution can be placed in an evaporating dish and left in a warm place for several days to allow the water to evaporate slowly. This method produces large crystals of sodium chloride, which appear as white cubes.
- Place the sodium chloride solution in an evaporating dish and heat slowly over a Bunsen flame to boil off the water. This is much faster, but the sodium chloride will not form noticeable crystals.
Any heating should be carried out wearing goggles and on a heat-proof mat. Note that when heating saline in an evaporating dish the salt can “spit” out of the dish as the water level gets low. The salt ejected in this way is very hot and will burn on contact with the skin or eyes. Remove thr heat source if they happens and allow the remaining water to eveapoarte without further heating.