Improving Processes and Products
Extracting Metals
Metals are very valuable materials but only a small number of them can be found as pure metals in the earth. The more reactive metals such as zinc, iron, copper, aluminum etc are found as metal ores. An ore is a rock containing a high enough quantity of a metal compound to be commercial useful. Metals as an ore are in the form of a compound, often with silicon, sulfur or oxygen.
Any metal that is lower than carbon in the reactivity series can be extracted from its ore by reacting it with carbon or carbon monoxide.
Iron is found in the form of haematite (Fe203) and zinc is mined as a mineral of zinc sulfide, copper is found in many forms including as copper oxides.
Zinc
Zinc can be extracted from zinc sulfide by heating in the presence of oxygen to form zinc oxide and then heating with carbon, the carbon is more reactive than the zinc and so it reduces it to zinc metal forming carbon dioxide.
Zinc sulfide + oxygen → Zinc oxide + Sulfur dioxide
2ZnS + 3O2 → 2ZnO + ↑ 2SO2(g)
Zinc oxide + Carbon → Zinc + Carbon dioxide
2ZnO + C → 2Zn + ↑ CO2(g)
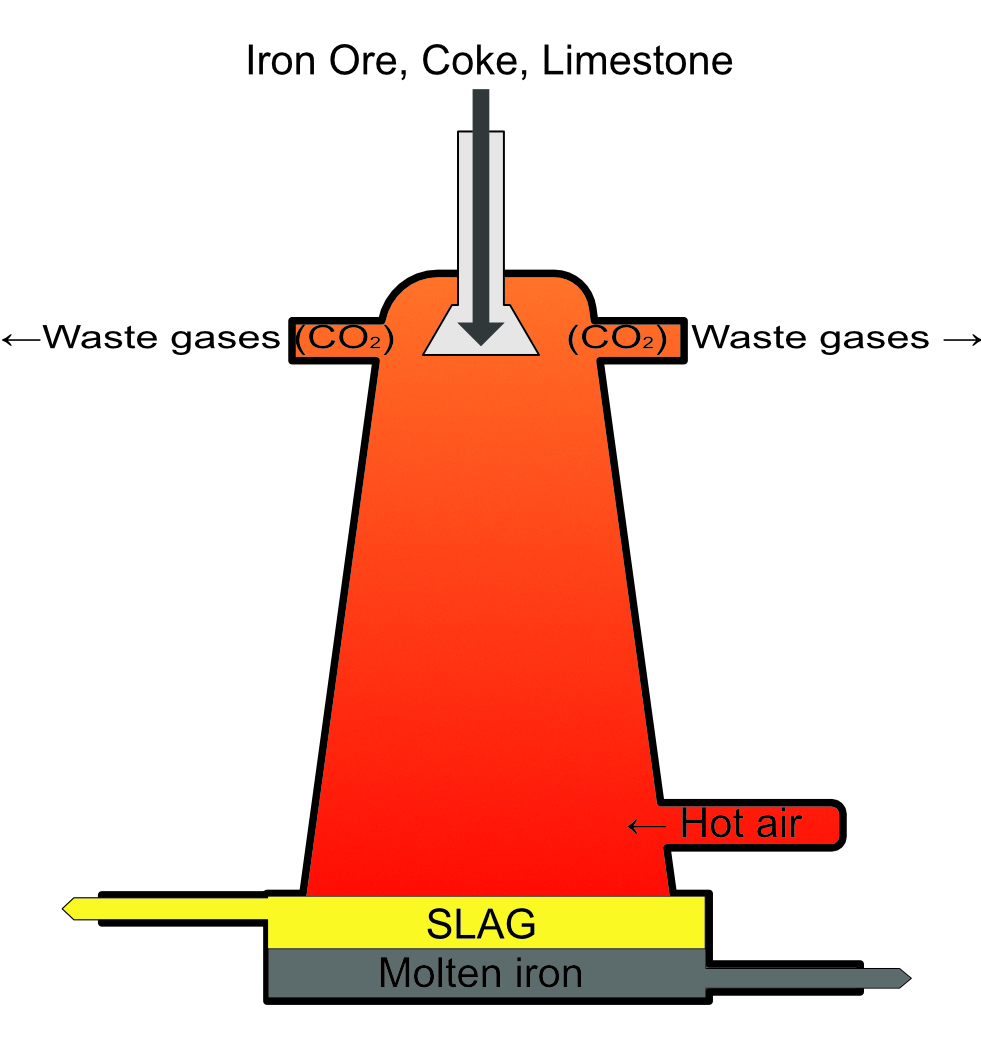
Iron - Blast Furnace
A blast furnace converts the iron ore haematite to iron metal and removes the impurities from the ore which are mostly sand and rock consisting of silicate SiO2.
Ingredients: Iron ore (Fe2O3), Limestone (CaCO3) and carbon in the form of coke.
The term blast furnace comes from the fact that the ingredients are heating to a high temperature with a “blast” of superheated air, providing the energy for the reaction and the oxygen required for the processes.
Step 1: Carbon is partly oxidized to carbon monoxide
2C + O2 → 2CO
Step 2: Iron(III) oxide is reduced to iron by the carbon monoxide
Fe2O3 + 3CO → 3Fe + ↑ 3CO2(g)
Removal of silica impurities, if this was not removed the resulting iron would be very brittle.
Step 1: Limestone (CaCO3) is converted to calcium oxide and carbon dioxide by thermal decomposition.
CaCO3 + heat → CaO + ↑ CO2(g)
Step 2: Calcium oxide reacts with the silicon dioxide to form calcium silicate (SLAG).
CaO + SiO2 → CaSiO3
The waste carbon dioxide is vented to the atmosphere. The SLAG and the molten iron sink to the bottom of the furnace, the iron is more dense than the SLAG so it sinks to the base and the SLAG floats on top of it. They can then both be pumped away separately.
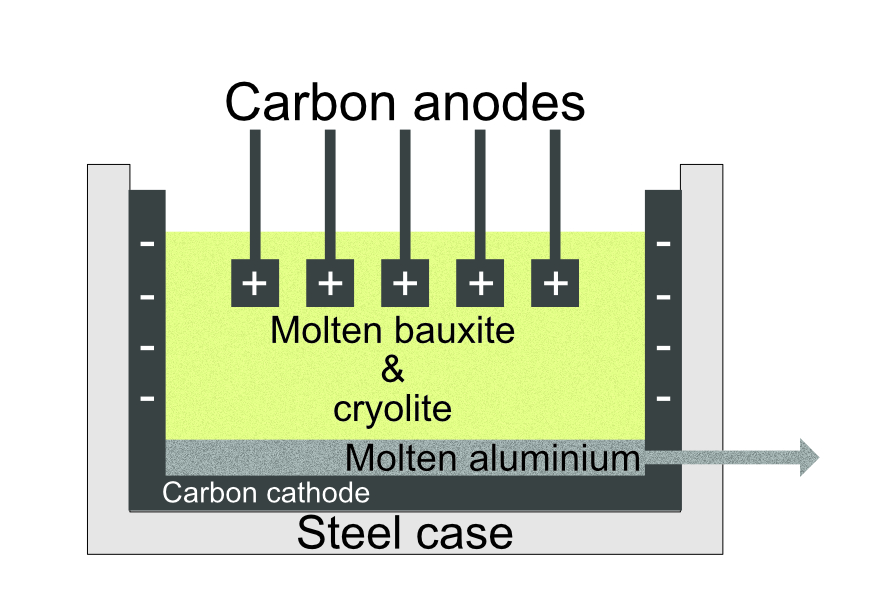
Electrolytic Extraction
Metals that are too reactive to be extracted by heating with carbon can be extracted from their molten ores by electrolysis. However, this requires a large amount of energy to melt the ore and to provide the high voltage required for this process.
Although Iceland has no natural aluminum ore (bauxite) they are one of the largest producers of metal aluminium. They import bauxite from Australia etc and process it in a large factory just outside the capital city Reykjavik. Iceland gains virtually all its electricity from geothermal power, this makes the electricity used for the extraction extremely cheap, unlike other countries that have to burn expensive fossil fuels or use nuclear power. This makes the Icelandic process both cheaper and more sustainable as it uses a renewable energy resource.
Bauxite must be in a molten form for the electrolysis to work. Bauxite melts at well over 2000oC so it is mixed with cryolite, this reduces the melting point and helps to reduce the energy needed to make aluminium metal.
At the cathode: The positive aluminium ions gain electrons and convert to aluminium atoms. The aluminium is molten at this temperature and can be pumped out.
At the anode the oxide ions lose electrons and become oxygen atoms, these react with the carbon electrodes to form carbon dioxide which bubbles off to the atmosphere.
Life Cycle Assessment
The life cycle of any product consists of the following stages:
- Obtaining and processing the raw materials
- Manufacturing or construction
- Transport of raw materials and finished products to market and point of use
- Use of the product
- Disposal, recycling or reuse of product
During each of these phases the environmental impact is considered, this includes;
Materials used, including water for cooling etc
- Minerals from mining, such as metal ores etc
- Crude oil / natural gas used a materials rather than energy (naptha for plastic etc)
- Water from groundwater, rivers, lakes and desalination of sea-water
- Gases distilled from the air
- Recycled materials
Energy consumption
- From fossil fuels (coal, oil and gas)
- Renewable sources
- Nuclear energy
Release of waste chemicals
- Carbon dioxide released into the air
- Sulphur dioxides / nitric oxides from fossil fuels released into the air
- Water pollution
- Heavy metals released into water or soil - mercury etc released from the production of magnets for wind turbines, for example
- Toxic byproducts
Land use implications
- Deforestation
- Habitat destruction
- Mining
- Creation of roads, airports, ports, factories and housing etc for workers
- Landfill and reclamation sites
- Expansion of commercial factory farming and large scale monoculture agriculture
- Waste processing plants including sewage treatment works.
By considering these four factors during each of the five phases of the products life cycle it is possible to estimate the effect on the environment of a product. In any assessment the impact is only an estimate as it is often impossible to fully appreciate all the effects of the life cycle of a product on the environment.
Biological Extraction
The traditional means of extracting metals via heating with carbon, smelting and by electrolysis using high grade metal ores is becoming increasingly difficult and expensive as reserves of these ore become overused. With an ever increasing population and the demand for metals rising for construction, manufacturing and the provision of electrical cables, the traditional reliance on high grade ores is no longer sustainable.
There are low grade ore, which can include seawater. These contain valuable metals but in amounts that are too low for conventional extraction. Many plants and bacteria naturally take up these metals as mineral salts from soil and water. They do not use them in their bodies so they accumulate (build up) to high concentrations.
Phytoextraction
Plants can be grown in soil or in water that contains low grade metal mineral salts. The plants are harvested and then burnt. The remaining ash contains a higher concentration of the metal than the soil or water and these remains can be processed to produce a pure metal.
Bacterial extraction - or bioleaching
Bacteria can be used to extract metal compounds from solutions, the bacteria convert the metals to salts that are often acidic. This is one potential problem with this process if it is not contained. The resulting acidic solution can then be processed using electrolysis or reacting it with carbon to extract the metals.
Recycling
Recycling is often confused with reusing and re-purposing. Recycling involves the processing of a material or product to obtain the useful materials from it so that they can be used to manufacture a new product. Reuse is when a product is used again for the original purpose, normally after some renovation or cleaning. Re-purposing is when a product is adapted for a new use.
Glass is melted down and made into new bottles - recycling.
Bottle cleaned and made into a lamp - re-purposing.
Milk bottles cleaned and refilled with milk - reusing.
Recycling requires material and energy. The original product once it has been used must be collected, stored, sorted, processed to obtain the useful material(s), manufactured into a new product and then transported for use.
The decision as to whether a material will be recycled is a commercial and technical one.
These factors are normally consider:
- How limited is the material, is the natural resource in limited or short supply?
- How much of the material is available for recycling, does it exist in commercially viable amounts?
- How much will it cost to reclaim and recycle?
- Can the impurities be removed easily to leave a pure enough product?
- How much will the energy cost for transport and processing compared to extracting the material from new?
- What are the environmental consideration?
- What demand is there for the recycled product and will it sell for a profit?
As materials become more rare and the world’s limited supplies of ores and fuels get low, more materials will be economically worth recycling and the technological costs will be economically viable.
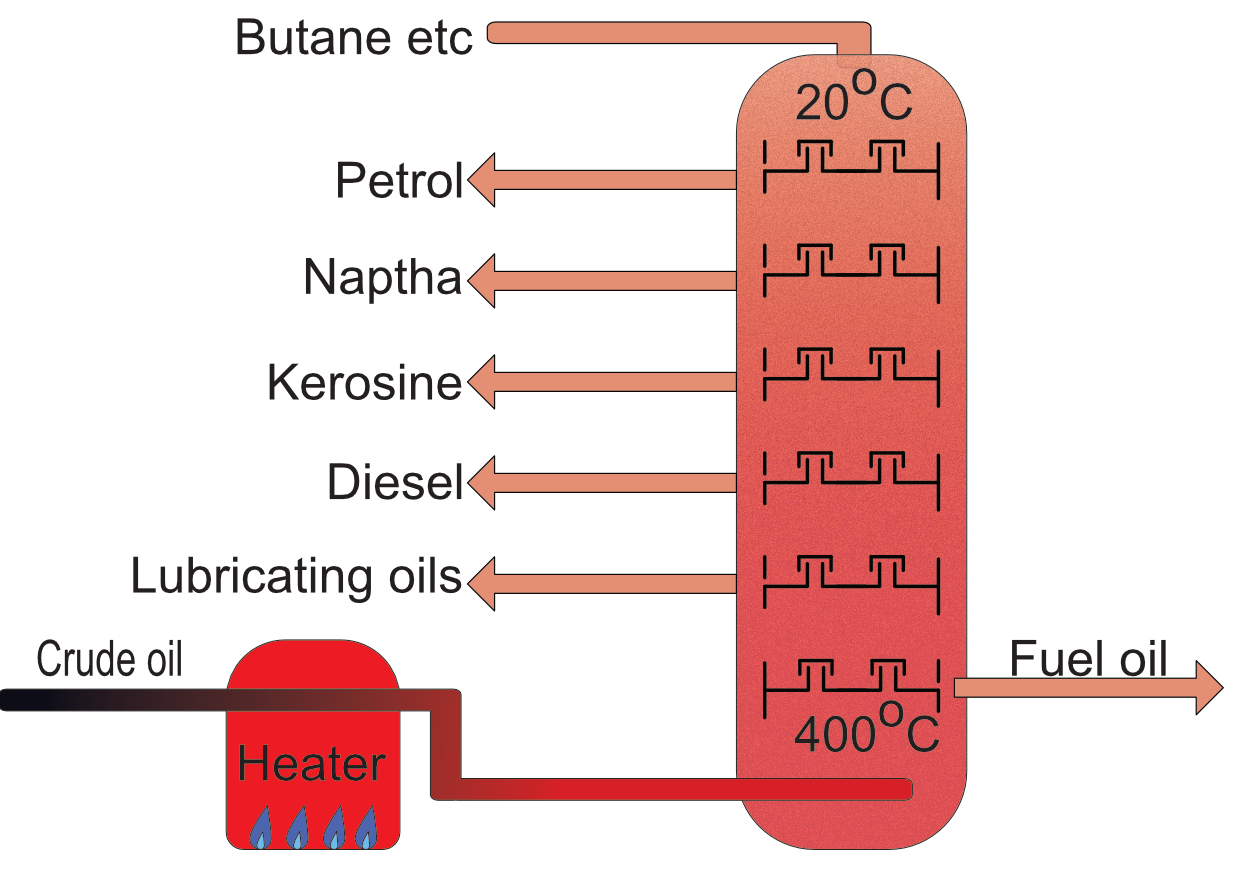
Crude Oil
Crude oil is the term given to the mixture of hydrocarbons extracted from the ground by drilling. This mixture is very thick and until processed has no applications. It is valuable because it contains hydrocarbons such as petrol, diesel, kerosene, aviation fuel, naptha for plastics etc.
The hydrocarbons in crude oil mainly belong to one group of chemicals called the alkanes. They contain only hydrogen and carbon with the general formula CnH2n+2.
- Methane CH4
- Ethane C2H6
- Propane C3H8
- Butane C4H10
This sequence includes octane, C8H18 an important component of petrols.
Each alkane has a different boiling point, increasing as the molecule gets larger, the first five are all gases at room temperature and are used in bottled gas for camping stoves etc. Octane has a boiling point of 125oC so is liquid at room temperature.
The differences in boiling points of the alkanes that make up crude oil can be used to separate them into useful products using fractional distillation.
Many of the fractions are used as fuels, some, notably naphtha, is used in the petrochemical industry for the production of plastics.
Other heavier fractions made of larger molecules are used for tar on roads, bitumen for protecting wood and as lubricating oils in engines etc.
The modern world and its economy has become heavily dependant on the use of products from crude oil, however, it is a non-renewable resource and some estimates suggest that the world only has around 50-70 years supply left. This gives countries with oil reserves a valuable commodity and considerable political and economic power.
Cracking
The mixture of alkanes that make up crude oil differs in each part of the world and many contain larger alkanes that are less useful. These larger alkanes can be split using heat and high pressures to produce smaller alkanes of more commercial use and value, plus a different hydrocarbon family called an alkene (CnH2n). This process is called cracking. Alkenes are a valuable product that can be used to make other products including plastics such as polyethene etc.
Example
Decane (C10H22) → Octane (C8H18) + Ethene (C2H4,)
Decane has a limited number of uses, but octane is used to manufacture petrol and ethene is used to make polyethene which is used in the production of plastic food wrappings, plastic bins, drinks bottles and even water pipes etc.
Although cracking crude oil fractions requires a lot of energy it produces a range of more useful fractions both as alkanes and alkenes for fuel and manufacturing.
During cracking a long chain alk__a__ne is converted to a shorter chain alk__a__ne and an alk__e__ne.