Distillation
Distillation
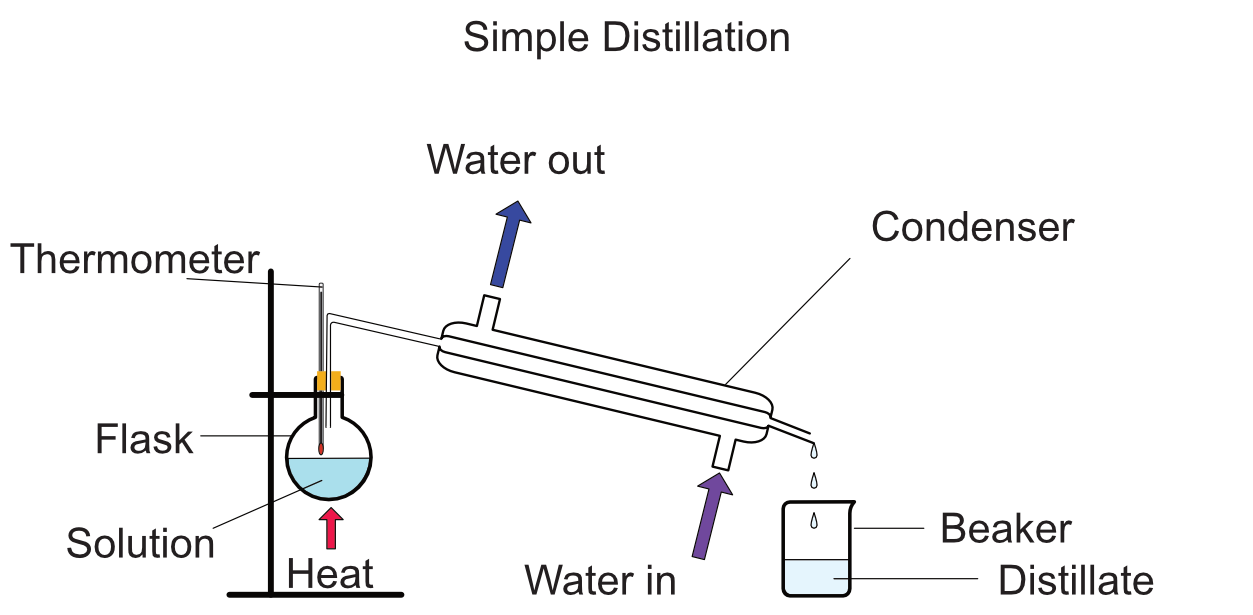
Distillation is used to seperate a mixture of liquids to obtain one or more of the chemicals in the mixture. Distillation works by heating the mixture to the boiling point of one of the chemicals. Starting with the one with the lowest boiling point. The gas produced is collected, feed into a condensing tube that cools the gas and changes it back to a liquid. This can then be collected at the end of the condensing tube.
The condenser is a tube with a hole through the middle, surrounded by a moving stream of cold water. The gas passes through the middle and the water cools it making the gas condense back to a liquid.
Safety: Wear goggles during heating. Use gloves to handle apparatus at the end, it will be hot.
Set-up
Remember when heating any glassware with a bunsen burner to start with the air hole half open, gently heat the glass until warm then if required the air hole can be fully opened. In this experiment, it is normally best to use a bunsen with the air hole half open to allow greater control of the temperature. The bunsen should be removed when the thermometer is 10oC below the boiling point of the chemical to be collected. The residual heat in the glass will continue to heat the solution. Never use a bunsen burner with the air hole closed for heating. This yellow flame is the safety flame and is not used for heating.