Halogens
Physical Properties
The halogens make up group 7 of the periodic table, they are all non-metals.
Properties
- Found as molecules rather than individual atoms, F2,Cl2, Br2 etc
- Distinctive colours, chlorine green, bromine brown, iodine purple.
| Element | Melting pointoC | Boiling pointoC | State at room temp |
| Fluorine F2 | -220 | -188 | gas |
| Chlorine Cl2 | -101 | -35 | gas |
| Bromine Br2 | -7.2 | 58.8 | liquid |
| Iodine I2, | 114 | 184 | solid |
| Astatine At2 | 302 | 337 | solid |
The melting points and boiling points increases moving down the group. This is due to the increase in the mass and size of the atoms and the increasing intermolecular forces between the halogen molecules.
They all have strong antiseptic properties, chlorine is used to sterilize drinking water and water in swimming pools, iodine is used in medicine for killing bacteria on cuts etc.
Chemical Properties
The term halogen means “salt making”, all members of this group form salts when reacting with metal elements. Sodium chloride, NaCl, is probably the best known of all the salts.
Sodium + Chlorine → Sodium chloride
2Na + Cl2 → 2NaCl
The elements become less reactive moving down the group, fluorine and chlorine will react with metals with little or no heating, iodine will react with metals but normally requires heating with a bunsen burner to start the reaction.
Halogens react with hydrogen to form halides, these form acidic solution when dissolved in water.
H2 + F2 → 2HF
H2 + Cl2 → 2HCl
- Fluorine forms hydrogen fluoride, HF and dissolves to make Hydrofluoric acid, HF(aq)
- Chlorine forms hydrogen chloride HCl and dissolves to make hydrochloric acid HCl(aq)
This pattern continues for the rest of the group.
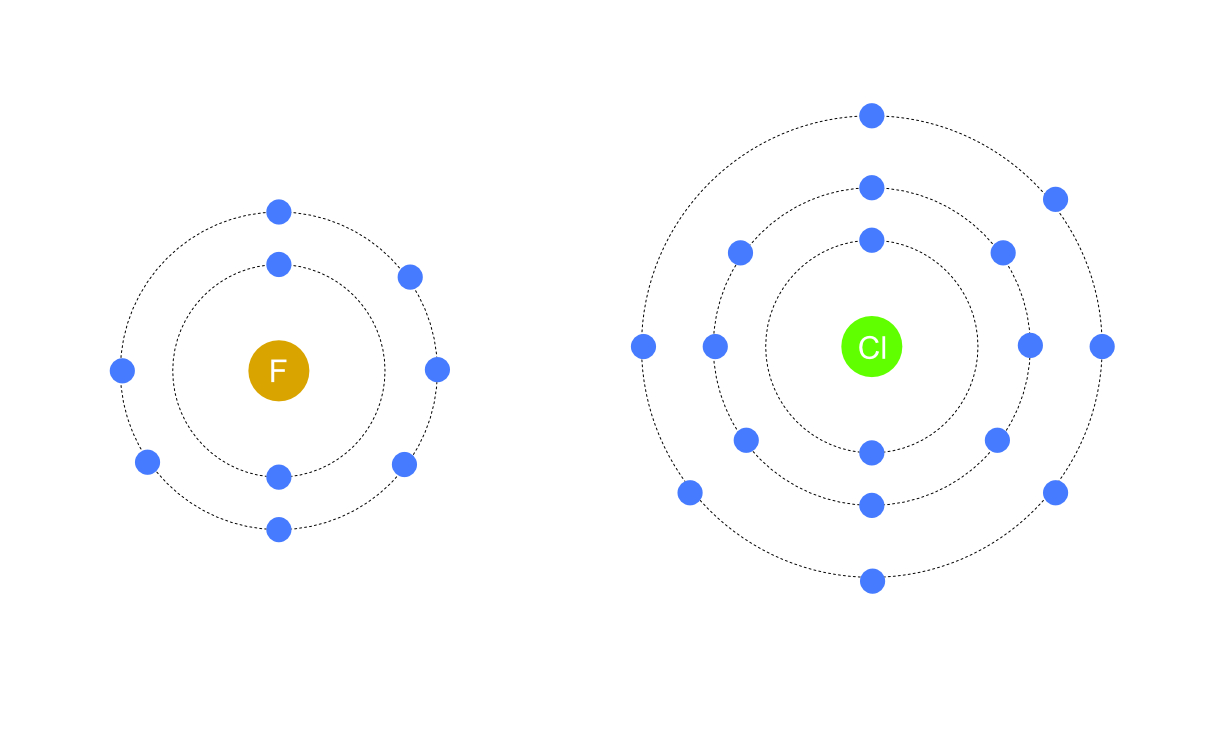
Electron Structures
The halogens form group 7 of the periodic table, this means that they all have 7 electrons in their outer orbit. This controls the nature of their chemical reactions, for example they all form ionic bond with group 1 in which an electron from the metal is taken into their outer orbit to form a negative ions with a charge of -1.