History of an Atom
Before the Electron
Our understanding of the atomic model has changed throughout ages:
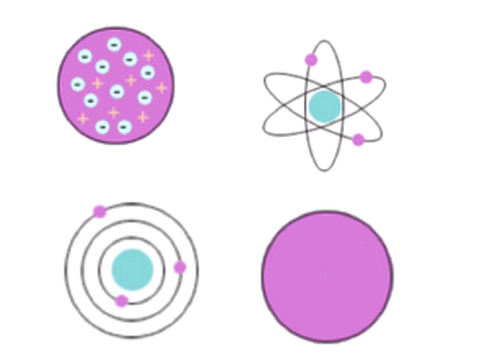
At first, before the discovery of the electron there was Dalton’s model. Here, atoms were thought to be tiny spheres that could not be divided.
Plum Pudding Model
The discovery of the electron forced an update in the model. The new model was developed by a scientist called Thomson, but the model itself is often called the plum pudding model.
This model suggested that an atom was a sphere of positive charge with negative electrons embedded within.
Nuclear Model
There was a major change to the atomic model by another scientist called Rutherford. This new model is called the nuclear model.
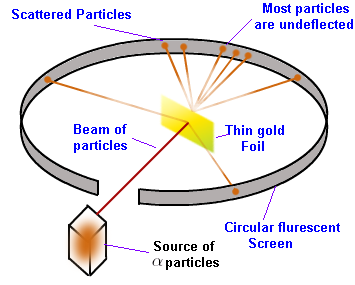
He conducted an experiment now known as the alpha particle scattering experiment (set up shown below). He fired a beam of alpha particles (Helium nuclei) at a very very thin stripe of gold foil. A number of observations led to some new conclusions and insights.
The observations and conclusions were as follows.
It was originally expected that all the alpha particles would pass through the foil without any deflections.
After the Nuclear Model
Not long after, another scientist (Bohr) and student to Rutherford, updated the nuclear model. He calculated that electrons do not orbit the nucleus like planets around the sun, rather electrons orbit at very specific distances from the nucleus. These distances were known as different energy shells.
Neutron Discovery
A few decades later James Chadwick discovered the neutron and that is resided in the nucleus. This then led to the current atomic model, the quantum cloud model. You do not need to know about the quantum cloud model at GCSE level, but it is worth knowing that the atomic model story continues into KS5 and beyond.
This topic lends itself really easily to four and six mark questions. Make sure that you are able to confidently describe the differences between each of the models and how the nuclear model replaced the plum pudding model