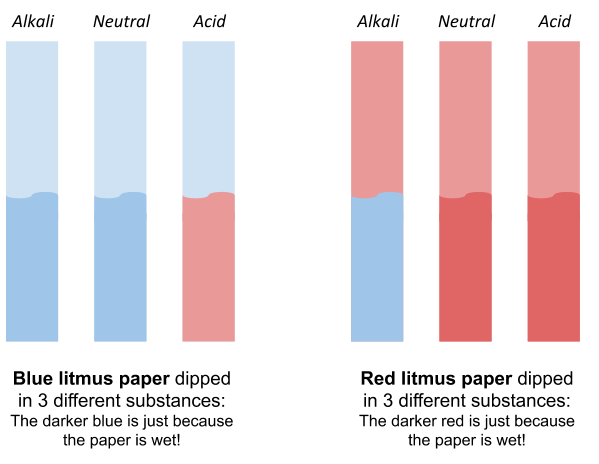Acids and Alkalis
The PHScale
What you need to know:
What the pH scale is
The different ways in which to test where a substance lies on the pH scale.
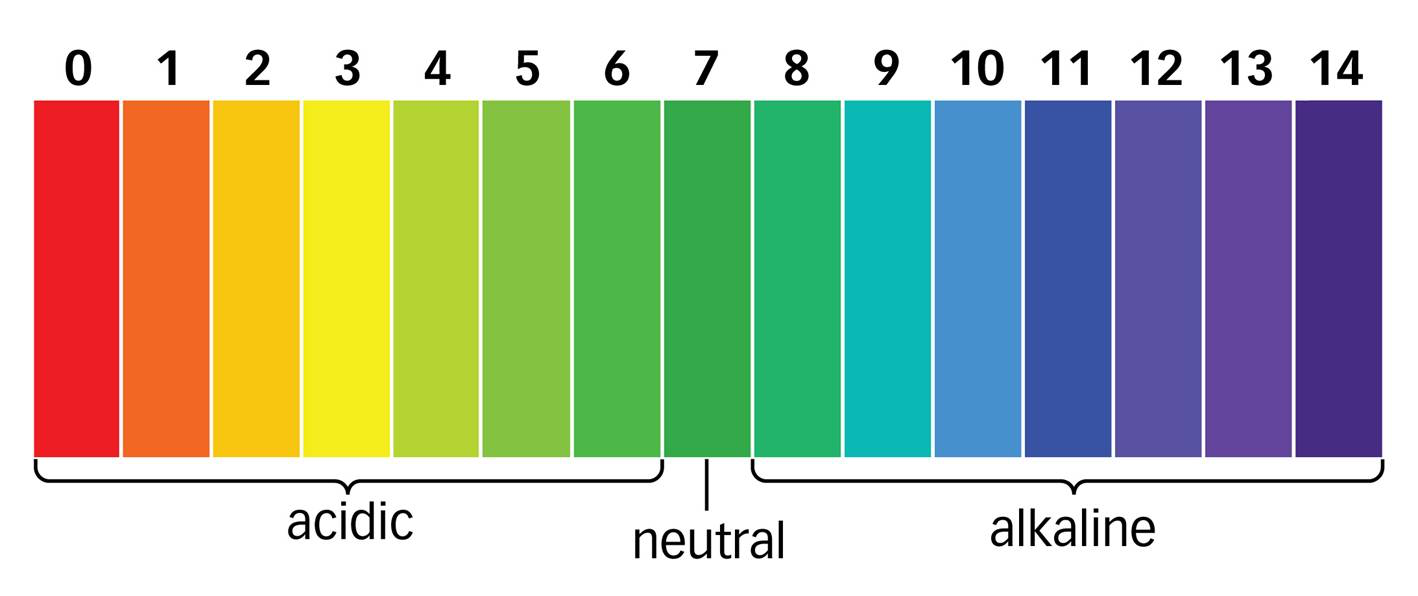
The pH scale is a scale to measure how acidic or alkaline a substance is. The scale ranges from 0 (very acidic) to 14 (very alkaline), with 7 being neutral. Note that another word for ‘alkali’ is ‘base’.
Testing pH
One way to test the pH of a substance is with an indicator.
There are lots of different types of indicators, but you will need to know about:
- Universal indicator
- Litmus
- Phenolphthalein
- Methyl orange.
Another way to test the pH of a substance is by using a pH probe to test the pH electronically. This gives a more accurate result than indicators.
For now, let’s just talk about universal indicator and litmus.
Universal indicator
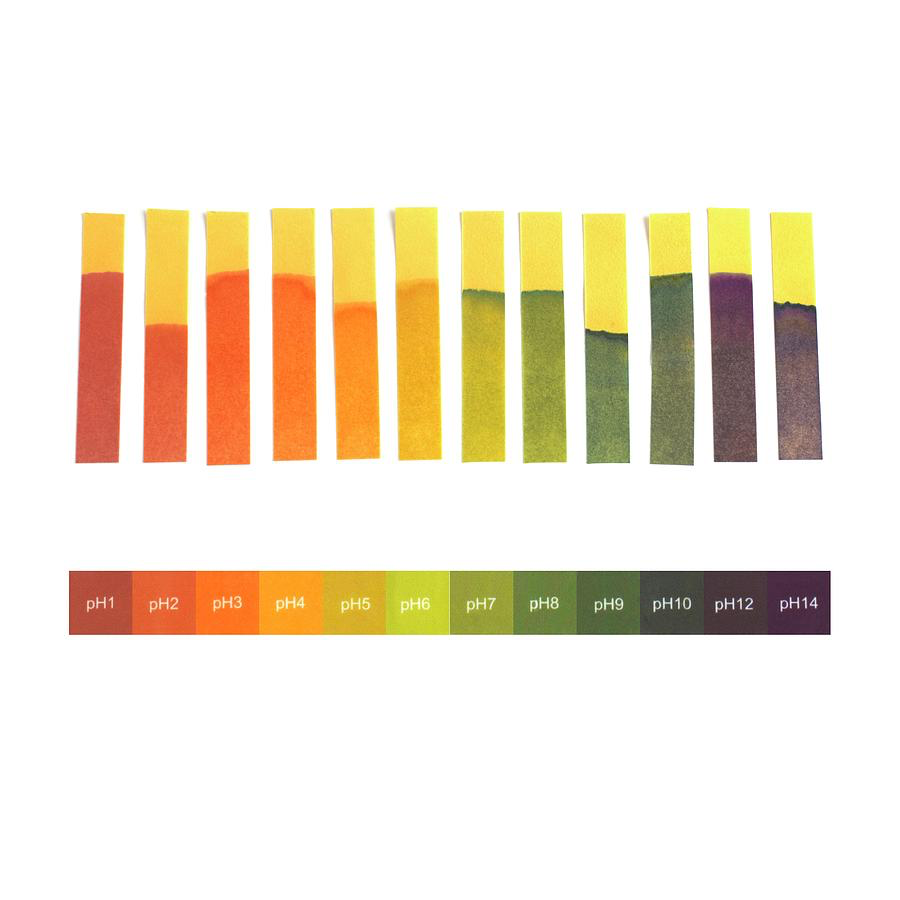
Universal indicator can be in paper form or as a liquid dye. The paper form starts off as yellow but turns colour when put into a liquid. The pH of the liquid determines what colour the paper will turn.
The same happens for the liquid indicator, the liquid indicator turns colour depending on the pH of the substance it is put into.
Litmus
You will usually see this in paper form. There are 2 types of litmus paper, red litmus paper and blue litmus paper. You will need both types to test a full range of solutions. Here is what happens to each type of litmus paper when dipped into different types pf solutions.
Litmus is not as useful as universal indicator, since it doesn’t tell you how acidic/alkaline a substance is, just if it is acidic or alkaline.
Acids and Alkalis
What you need to know:
- What the difference is between acids and alkalis.
- What ions are responsible for making something acidic or alkaline.
- The difference between strong and weak acids.
- How acids and alkalis can neutralise one another.
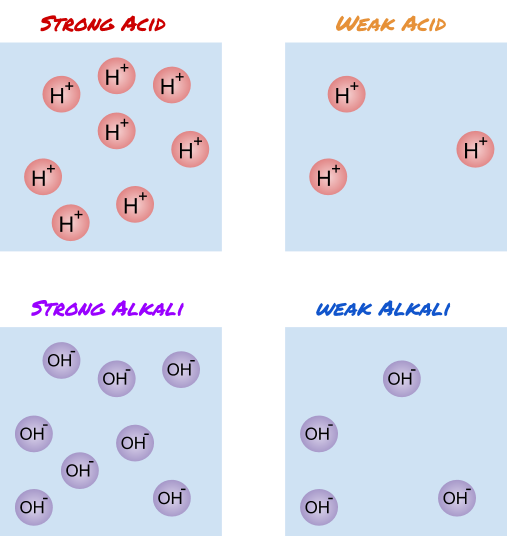
When something is acidic, it means that there are H+ ions released when the substance is dissolved in water.
When something is alkaline, it means there are OH− ions released when the substance is dissolved in water.
The amount of ions in the acid or alkali determines how strong it is. For example, the more H+ ions in the acid, the stronger the acid is.
Note: Each __OH−_ ion is one oxygen atom and one hydrogen atom bonded to make one particle. There is an extra electron between them._
Each H+ ion is just one atom of hydrogen with one less electron than it should have.
Neutralisation reaction:
When an acid and alkali react together, they produce a salt and water:
acid + alkali → salt + water .
This is because the ____H+ __</sup>__ions (from the acid) and the ____OH−__ ions (form the alkali) come together to form water:__
As you may know, water is neutral, so the acid and the alkali have come together to neutralise each other (to make a pH 7).
Since the strength of the acid and the alkali are not the same, you will need different amount of each for the two to neutralise (there needs to be the same amount of H+ and OH− ions to fully neutralise).
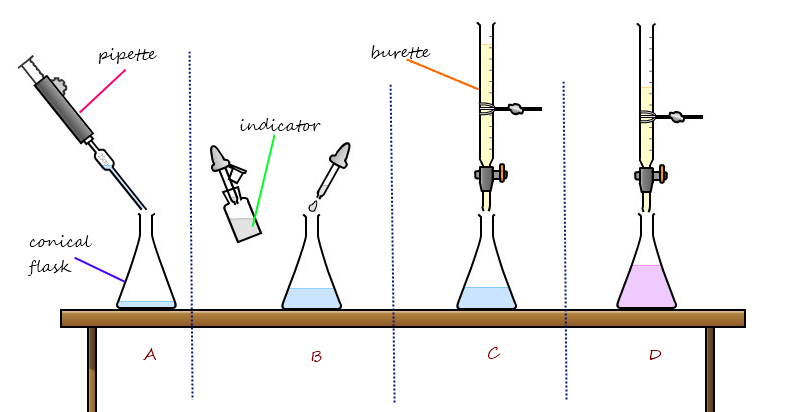
Titration Equipment
What you need to know:
How to carry out a titration reaction.
What the different pieces of equipment are used for in titration reactions.
How to properly read a burette
A titration experiment is used to find out how strong an acid (or an alkali) is. This is done by neutralising an acid with an alkali/base. If you know the strength of one of them, you can work out the strength of the other.
There are three main pieces of equipment needed for a titration.
Burette: This is used to hold an acid or alkali and release it from the bottom, drop by drop. There is a tap on the bottom of the beurette to stop/start the release of the liquid. The burette has measurement markings so that you can see how much liquid has been released.
Pipette: A pipette is used to precisely measure out an amount of liquid and drop it into the conical flask. The pipette is more precise than the conical flask since it is very thin.
Conical flask: This is just a glass container with a narrow neck. The narrow neck is essential to prevent any liquid from splashing out of the flask.
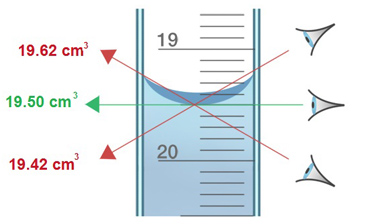
Reading a burette
Burettes can be quite tricky to read, so here is a quick guide on burette reading!
- Read the burette from the meniscus. When liquid sits in a burette it forms a little dome. You need to take your reading from the bottom of the dome. Make sure you’re looking head on to do this!
- The measurement markings on a burette start at the top, instead of at the bottom. See the picture above, 19 is above 20! This means that, to find the volume of liquid gone, you need to minus the initial reading from the final reading. I.e. if you started with a reading of 13.00cm3and you ended up with 19.50cm3, the volume that had left would be 19.50cm3- 13.00cm3= 6.50cm3.
Titrations
What you need to know:
How to carry out a titration reaction.
If you know the strength (concentration) of the acid or base, then you can work out the concentration of the other. This is done by seeing what volume of acid is needed to neutralise a set volume of the other.
__Here is a step by step guide for titrations: __
- Place the burette in the clamp and stand, about 15 cm above the table.
- Measure out a known volume of acid with the pipette (usually 25d㎥).
- Carefully place this into the conical flask with a few drops of indicator.
- Use a funnel to carefully fill the burette with alkali with known concentration. Run a little bit through the tap, in order to wash out any unwanted liquids from the tap.
- Place the conical flask underneath the tap of the burette. Lower the burette to ensure that the tap is just inside the neck of the conical flask. This avoids splashing.
- Create a rough titration by running the alkali into the acid whilst someone swirls the conical flask. The conical flask needs to be swirled to make sure the liquids are mixing well.
- As soon as the indicator shows neutral, stop the tap and record how much alkali had left the burette.
- Safely dispose of the mixed acid-alkali solution in the conical flask and thoroughly rinse with pure water. Fill the burette again with alkali. Fill the flask with acid again, using the pipette.
- Now you will record your three actual results. Do exactly the same thing of running the alkali into the acid (whilst someone swirls). When the liquid approaches your rough titration, stop the tap and drip in the alkali drop by drop.
- As soon as the indicator shows a neutral result, stop the tap completely and record how much alkali was needed.
- Repeat steps 8-10, at least, two more times.
- Calculate a mean volume from your results. This will give you the mean amount of volume needed to neutralise a set volume and concentration of acid.
Indicators
What you need to know:
What different indicators are used to test for pH.
The main indicators you need to know about are:
- Universal indicator
- Litmus
- Phenolphthalein
- Methyl orange.
We have already spoken about Universal indicator and litmus paper in the ThePHScale section.
_Phenolphthalein, methyl orange and litmus are known as single indicators. This is because they only contain one compound that changes colour at a particular pH. This means that they are particularly good for titrations (but terrible for seeing how strong an acid/alkali is). _
Universal indicator is known as a wide range indicator. Wide range indicators are good at estimating the pH of a solution, but cannot be used in titrations. The colour change is too gradual for a titration (a really weak acid/alkali has a very similar colour to a neutral solution).