Alcohols and Carboxylic Acids
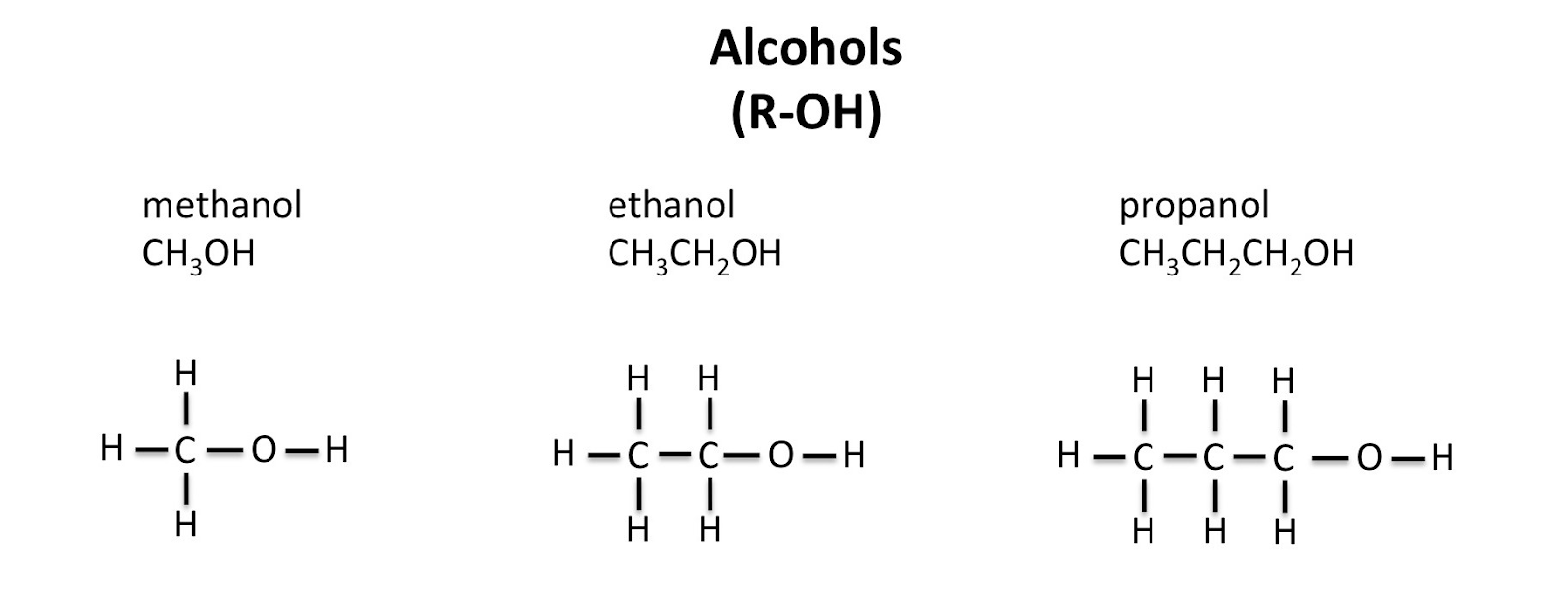
Alcohols
Alcohols are a group of compounds that have a hydrocarbon chain attached to an -O-H group. The general formula for an alcohol is 
The___ -O-H___ part is what makes it an alcohol!
Properties of alcohols
- Flammable
- The first four (so methanol, ethanol, propanol and butanol) alcohols dissolve completely in water to make a neutral solution.
- They react with sodium
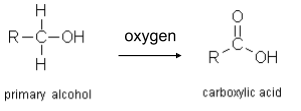
- They form carboxylic acids when they react with oxygen (from the air). More on carboxylic acids later.
Uses of Alcohols
__Alcohols have many uses: __
- Alcoholic drinks - Ethanol is mainly used. Methanol can blind you if drank. Alcohol poisons the body, which makes people ‘drunk’.
- Solvents - alcohols can dissolve most substances that water can dissolve and more.
- __Fuels __- since alcohol is flammable, it can be burnt with air which releases energy.
Making ethanol from sugars
Yeast turns sugar into carbon dioxide and ethanol. This is one of the main ways that ethanol is produced industrially. This is called fermentation.
In lots of alcoholic drinks, the alcohol isn’t added but it’s made in the liquid by adding yeast. The yeast turns the sugar into carbon dioxide gas and ethanol!
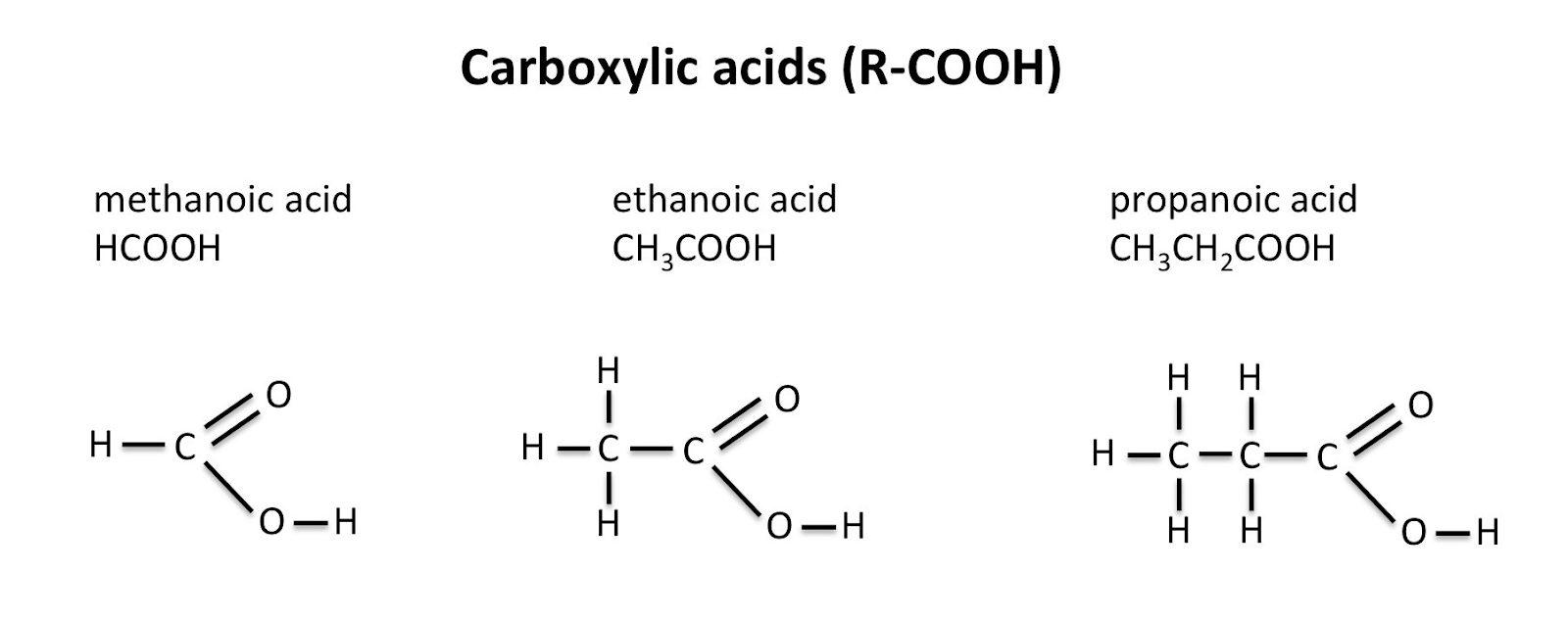
Carboxylic Acids
Carboxylic acids have a -COOH group added to the hydrocarbon chain. These are named the same way as alkanes and alkenes, except we write ‘-anoic acid’ at the end instead of -ene or -ane.
Properties of Carboxylic Acids
Properties of carboxylic acids
- They react like acids (for example, they neutralise bases)
- Dissolve in water
- They are weak acids - they only partially ionize in water (see #StrongAndWeakAcids2 section).
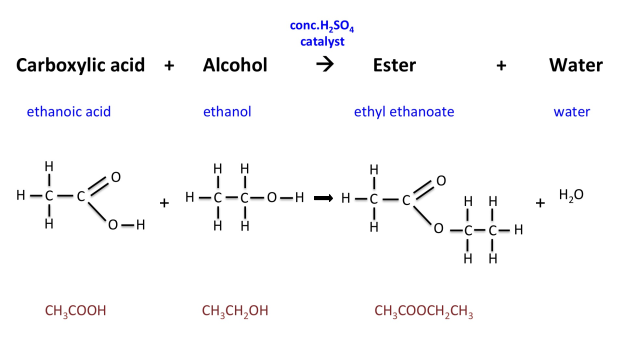
- They can react with alcohols to form esters
Esters
Esters are formed when an alcohol reacts with a carboxylic acid.