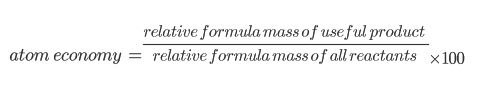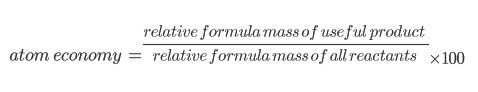Atom Economy and Yield
Atom Economy
A lot of chemical reactions make more than one product. Not all of these products are useful. We can calculate how efficient a reaction is by finding out what percentage of the reactants actually turned into useful product, using the equation below!
If there is 100% atom economy, it means that ALL of the reactants have been turned into useful products. It is cheaper and more economical to have a high atom economy. Companies don’t want to pay for lots of raw materials to not get much out in return.
The best way to to get around this is to find a use for the other products (so that they are no longer waste)!
Any products that are not counted as useful, are counted as waste. You can also find the percentage waste of something. To do this, you just need to divide the waste product by the reactants and x100.
Percentage Yield
The theoretical yield of something is the amount of product that could have been made if none of the products were lost and if all of the reactants actually reacted.
Reactions don’t always happen exactly as we think they should.
There are a few reasons that a reaction might not fully react:
Atom Economy Calculations
Example 1:
Calculate the atom economy of the following reaction to produce Hydrogen:.
Answer:
Step 1: Find the of the reactants and of the useful product.
Step 2: Now find the atom economy by using the equation.
Example 2:
Calculate the atom economy of the following reaction to produce magnesium oxide:.
Answer:
Step 1: Find theof the reactants and of the useful product.
Step 2: Now find the atom economy by using the equation.
Percentage Yield Calculations
The theoretical yield of something is the amount of product that could have been made if none of the products were lost and if all of the reactants actually reacted.
Reactions don’t always happen exactly as we think they should.
There are a few reasons that a reaction might not fully react:
- Some reactants could be lost
- Some products could be lost
- The chemical mix could be contaminated with another chemical. The other chemical could react with some of the reactants to make an unknown product.