Condensation and Natural Polymers
Condensation Polymers
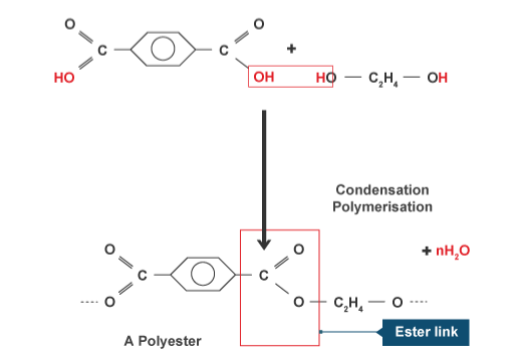
Condensation polymers occur when a ‘double ended’ carboxylic acid forms an ester bond with a ‘double ended’ alcohol.
This can happen with other functional group reactions also.
Since there is a functional group at each end, it means that there can be a ester bonds can form at the beginning and end of each molecule. Forming one long chain - a polyester!
Condensation vs Additional Polymerisation
  |
Naturally Occuring Polymers
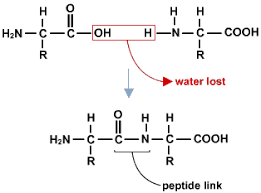
Amino acids can form long polymer chains called polypeptides. These amino acids are the monomers and they form __polypeptide __chains via a condensation reaction (just like the condensation polymerisation reaction, but slightly different).
The only difference is that there is an __amino __group instead of an alcohol group! It works exactly the same way though!
The link formed is not called an ester link, it is called a peptide link/
Proteins are made out of long chains of polypeptides.
Proteins have lots of important roles in the body:
- To make enzymes
- To make haemoglobin in the red blood cells
- To make antibodies for the immune system
- To make body tissue!
So, proteins are pretty important.
DNA
DNA is made of two long polymer chains connected down the middle by base pairs. The monomers that make up the polymer chains are called nucleotides.